Trump's Psychedelics Executive Order: What It Means for Magic Mushrooms and Mental Health (2026)
Publicado de: Psilocybin Science & News

Published April 21, 2026 — Psilocybin Science & News
In this article: On April 18, 2026, President Trump signed a landmark psychedelics executive order directing the FDA, DEA, and VA to fast-track research and access for psychedelic treatments — including psilocybin and ibogaine. We were on holiday last week, so here’s a clear, friendly catch-up on what the order says, why it matters, and what it means for the future of psychedelic medicine.
The psychedelics executive order signed on April 18, 2026, may be the single biggest federal policy shift in this space in decades. President Donald Trump put his signature to a document titled “Accelerating Medical Treatments for Serious Mental Illness” — and in doing so, the United States government gave its clearest signal yet that psychedelics, including psilocybin and ibogaine, belong in mainstream medicine. We were away for a few days, but this is too important to skip. Here is what the order actually does, why it happened now, and what comes next.
Why a Psychedelics Executive Order, and Why Now?
The executive order opens with a striking set of numbers. Over 14 million American adults currently live with a serious mental illness. More than 6,000 veterans die by suicide every year — a rate more than twice that of the general adult population. Standard treatments, including antidepressants and psychotherapy, simply do not work for a significant portion of these people.
Against this backdrop, the science on psychedelics has been building quietly but powerfully. The FDA has already granted Breakthrough Therapy designation to specific psilocybin compounds. Compass Pathways recently completed a successful Phase 3 trial for treatment-resistant depression. Johns Hopkins, Harvard, Stanford, and dozens of other world-class institutions are running trials. The evidence is there. What was missing was federal momentum — and this psychedelics executive order provides exactly that.
The political backstory is worth knowing too. Reports suggest that podcaster Joe Rogan texted President Trump directly about ibogaine research and its potential for veterans with opioid addiction and PTSD. The order was reportedly drafted in under a week. The signing ceremony included former Navy SEALs, veterans’ advocates, and ibogaine researchers standing alongside the President. This was a political moment as much as a policy one.
Five Directives: What the Psychedelics Executive Order Actually Does
The order contains five concrete directives. Let’s walk through each one in plain language.
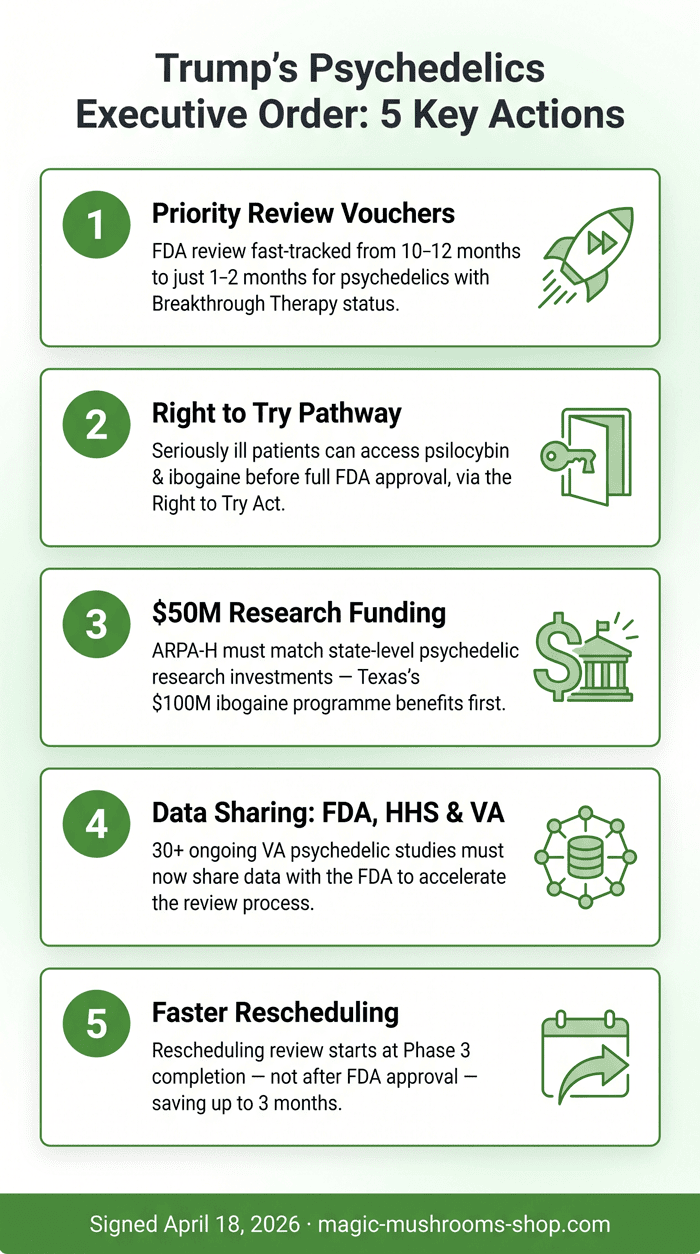
1. Priority Review Vouchers for Psychedelic Drugs
The FDA Commissioner must now issue Commissioner’s National Priority Vouchers to psychedelic drugs that hold Breakthrough Therapy designation. These vouchers dramatically speed up FDA review — from the standard 10–12 months down to a target of just one to two months. At the signing ceremony, FDA Commissioner Marty Makary announced that three new drug applications are already “imminent.” He also confirmed that clinical trial clearance has been granted for ibogaine, meaning US trials for that substance can now begin.
2. A Right to Try Pathway for Eligible Patients
The FDA and DEA must establish a pathway allowing patients to access investigational psychedelic drugs — including ibogaine — under Trump’s existing Right to Try Act. This opens a legal route for seriously ill patients to access promising treatments before full approval. Psilocybin is explicitly included. Legal experts at Harvard Law’s Petrie-Flom Center note this could benefit psilocybin access, which has been a point of ongoing legal contention.
3. $50 Million for Federal–State Research Partnerships
The Department of Health and Human Services must allocate at least $50 million through ARPA-H (the Advanced Research Projects Agency for Health) to match state-level investments in psychedelic research programmes. Texas is the most immediate beneficiary: the state recently committed $100 million to ibogaine clinical trials, and this federal matching fund brings serious new resources to that effort.
4. Data Sharing Between FDA, HHS, and the VA
The order requires the FDA, HHS, and the Department of Veterans Affairs to sign data-sharing agreements and pool clinical trial data. Approximately 30 psychedelic studies are already underway at VA sites — covering PTSD, depression in spinal cord injury, and alcohol use disorder. Until now, that data has not flowed freely into the FDA review process. This provision changes that.
5. Faster Rescheduling After Phase 3 Completion
The Attorney General must begin the rescheduling review process for any Schedule I substance that successfully completes Phase 3 trials — before FDA approval, not after. By initiating the review earlier, the order could shave off up to three months of post-approval delay. Psilocybin is currently Schedule I in the United States, which means it is treated as having no accepted medical use — a classification that has long blocked research and commercialisation.
| Signed | April 18, 2026 |
| Signed by | President Donald J. Trump |
| Official title | Accelerating Medical Treatments for Serious Mental Illness |
| Substances named | Ibogaine (explicitly), psilocybin (via Breakthrough Therapy designation), psychedelic drugs broadly |
| Agencies directed | FDA, DEA, HHS, ARPA-H, VA, Department of Justice |
| Funding | $50 million minimum via ARPA-H (existing funds) |
| Key mechanisms | Priority Review Vouchers + Right to Try pathway + rescheduling reform |
What the Psychedelics Executive Order Means for Psilocybin
You might be wondering: this order keeps mentioning ibogaine. What does it mean for psilocybin and magic mushrooms?
Quite a lot, as it turns out. While ibogaine gets the most explicit mentions — largely because of its strong support among veteran communities — the order’s language applies broadly to any psychedelic drug with Breakthrough Therapy designation. Psilocybin already holds that designation for treatment-resistant depression. That means it is directly in scope for the Priority Review Voucher programme.
As we covered earlier this year, Compass Pathways is preparing a rolling New Drug Application (NDA) to the FDA for its COMP360 psilocybin compound, with full submission expected in Q4 2026. The company responded enthusiastically to the executive order. With Priority Review Vouchers now in play, the review timeline after submission could shrink from a year to as little as six to eight weeks. An FDA-approved psilocybin medicine may arrive sooner than most people expect.
There is also the rescheduling provision. Psilocybin cannot be commercially sold or prescribed in the US as long as it sits in Schedule I. This psychedelics executive order instructs the Attorney General to begin the rescheduling process the moment Phase 3 is complete — not after the FDA decision. That is a meaningful procedural shift.
Want to understand how psilocybin works in the brain? Our guide on psilocybin and the mind covers the neuroscience in plain language — no chemistry degree required.
The Ibogaine Story: Veterans and a Plant From Africa
If psilocybin is the headliner, ibogaine is the surprise guest at this signing ceremony. Ibogaine is derived from the root bark of Tabernanthe iboga, a shrub native to Central Africa — particularly Gabon and Cameroon — where it has been used for centuries in the Bwiti spiritual tradition. In modern research, it has attracted attention primarily for its remarkable effects on opioid withdrawal and PTSD.
Multiple studies — many conducted in Mexico and other countries where ibogaine is legal — have shown that a single session can eliminate opioid withdrawal symptoms and create lasting reductions in PTSD symptoms in veterans. Former Navy SEALs have been vocal advocates. The problem until now has been cardiac risk: ibogaine can affect heart rhythm, which is why the FDA has historically resisted approving clinical trials. The psychedelics executive order explicitly names ibogaine and grants it a Right to Try pathway — an acknowledgment that potential benefit, for veterans with no other options, may outweigh the risk in controlled settings.
FDA Commissioner Makary confirmed at the signing ceremony that IND (Investigational New Drug) clearance has been granted for ibogaine — meaning US clinical trials can now legally begin for the first time. That alone is historic.
What Experts and the Industry Are Saying
Reactions from legal scholars, researchers, and industry have been largely positive but measured. Experts at Harvard Law’s Petrie-Flom Center point out several nuances worth keeping in mind.
An executive order is not self-executing. It directs agencies to act, but the FDA, DEA, and HHS retain their own legal processes. A president cannot compel rescheduling directly — that requires a full scientific and medical review. What the order does is send a strong political signal and remove internal bureaucratic resistance. Given that HHS Secretary Robert F. Kennedy Jr., FDA Commissioner Makary, CMS Administrator Dr. Oz, and NIH Director Bhattacharya all publicly supported the order, that signal is unusually unified.
From the industry side, Compass Pathways issued a supportive statement the following day. Law firm Orrick, which advises life sciences companies, called the order “a landmark policy development” while noting that real-world impact depends on agency follow-through.
One gap to note: The executive order says nothing about religious use of psychedelics. Ayahuasca churches and psilocybin spiritual communities, which have faced significant DEA pressure, are not addressed. Legal experts note this as a deliberate omission — the White House favours the FDA medical pathway over religious freedom or personal use routes.
How This Fits Into the Broader Psychedelic Landscape
This executive order did not arrive in a vacuum. It is part of a wave of developments we have been following here at Magic Mushrooms Shop. For context:
The US has been playing catch-up at the federal level. This psychedelics executive order represents the clearest signal yet that Washington is ready to move. Read our full 2026 psilocybin legalisation overview for a complete global picture. For the science behind these treatments, our in-depth guide on how psychedelics are transforming mental health treatment covers the clinical trial data in full.
What Comes Next — A Realistic Timeline
Here is an honest look at what to expect over the coming months.
| Milestone | Expected Timing | What to Watch |
|---|---|---|
| Ibogaine IND clearance | Already confirmed April 2026 | First US clinical trials can now begin |
| 3 NDAs “imminent” (FDA Commissioner) | Late April / May 2026 | Which substances? Likely psilocybin candidates |
| Compass Pathways COMP360 durability data | Q3 2026 | 26-week outcomes from Phase 3 trials |
| Compass Pathways full NDA submission | Q4 2026 | First-ever psilocybin NDA in history |
| FDA review (with Priority Voucher) | 1–2 months post-submission | Could mean approval as early as early 2027 |
| DEA rescheduling review (psilocybin) | Begins at Phase 3 completion | Psilocybin could move from Schedule I to II or III |
The road is not without obstacles. Psilocybin is still a Schedule I substance in the US and rescheduling requires formal scientific review that no executive order can override entirely. Reimbursement — whether insurance will cover these treatments — remains an open question. Delivery models also need resolving: is this a ketamine-clinic style service, or can a GP eventually prescribe it? These are real issues that will play out over the next two to three years.
But the direction is unmistakably clear. The United States government has just formally recognised that psychedelics have a role in treating some of the most difficult mental health conditions of our time. That is a profound shift — and one that was unimaginable just ten years ago.
Curious about how psilocybin helps with depression and PTSD? Our article on psychedelics and addiction treatment explores the clinical evidence across multiple conditions, including groundbreaking studies from Johns Hopkins and Imperial College London.

Curious about psilocybin? Explore our range of magic truffles — a legal, well-studied psilocybin experience, shipped from Amsterdam. Or discover our microdosing products for a gentler introduction.
Note: This article is written for educational and informational purposes only. Magic Mushrooms Shop does not encourage any activity that violates local, national, or international law. Psilocybin and ibogaine remain controlled substances in most countries. Always check the laws in your region before making any decisions.


April 21, 2026